Hypothesis / aims of study
Available data suggest that the extensive vascular plexus surrounding the female urethra has an important impact on urethral pressure and thereby on urethral continence mechanism. A potential treatment strategy for urinary incontinence associated with decreased urethral pressure could, therefore, be a pharmacologically induced expansion of this vascular plexus caused by local increase in arterial blood flow. Theoretically, vasodilation mediated by phosphodiesterase-type 5 (PDE5) inhibitor action could facilitate such expansion of the urethral vasculature. PDE5 expression has been reported in human female reproductive tissues, as well as in male genital and bladder tissue [1], and findings from a preclinical study in female rats indicate that PDE inhibition may increase baseline urethral pressure [2]. However, PDE5 inhibitors may also induce relaxation of urethral smooth muscle and the external urethral sphincter (EUS) thereby facilitating micturition. Accordingly, Datta et al. evaluated the PDE5 inhibitor sildenafil (50 mg twice daily for four weeks) in women with bladder outflow obstruction, but did not find statistically significant differences in voiding parameters compared to placebo [3]. Thus, the effect of PDE5 inhibition on the female urinary tract remains unclear. This study aimed at evaluating the effect of the long-acting PDE5 inhibitor tadalafil on opening urethral pressure (OUP) and voiding parameters in healthy women.
Study design, materials and methods
Healthy women were recruited for this double-blind, randomized, placebo-controlled, crossover study Participants were randomized to receive either single dose tadalafil (40 mg) or placebo at the first visit and then crossed over to the opposite treatment at the second visit. The visits were separated by a washout period of at least six days to avoid carry-over effects. Two hours after administration of study medication (at the reported time of peak plasma drug concentration), urethral opening pressure during resting and squeezing condition of the pelvic floor was measured using urethral pressure reflectometry. Immediately after urethral pressure measurements, uroflowmetry was performed with a prefilled bladder volume of 300 ml natrium chloride. We followed up on adverse events at the end of each clinic visit and by phone six days after the last visit. Based on an expected within-subject standard deviation of 5.4 cmH2O, we needed 24 women to complete the study to have a power of 99% to detect a 10 cmH2O difference in OUP between tadalafil and placebo with a significance level of 5%. We performed multiple regression analyses with subject, period, and treatment as covariates using SAS statistical software (v.9.4.6).
Results
Twenty-four women were included in the study, and there were no dropouts. Median age was 24.5 (range 20–43) years, and median body mass index 22.7 (range 19–26.9) kg/m2. Compared to placebo values, single dose 40 mg tadalafil caused a significant fall in resting OUP of 6.8 cmH20 (95% confidence interval [CI] 1.9–11.8, p=0.009) and of 8.8 cmH20 during squeeze (95% CI 3.1–14.6, p=0.005) (figure 1). Tadalafil did not cause any statistically significant change in voiding parameters compared to placebo (average flow rate: −0.8 ml/s [95% CI −2.0–0.4, p=0.2]; maximum flow rate −1.7 ml/s [95% CI −4.8–1.5, p=0.3]; voided volume −11.4 ml [−40.7–18.0, p=0.4]) (figure 2). We observed a statistically significant period effect for average flow rate (p=0.02) and maximum flow rate (p=0.02) with increased flow rates at the second visit compared to the first. Of a total of 41 adverse events, 36 adverse events were related to tadalafil treatment and five adverse events were related to placebo treatment. Headache was the most common adverse event (tadalafil: 15; placebo: 3). Most adverse events were mild in severity, and no serious adverse events occurred.
Interpretation of results
To our knowledge this is the first randomized, placebo-controlled study to assess the effect of the PDE5 inhibitor tadalafil on urodynamic and urethral pressure parameters in healthy women. Single dose tadalafil decreased OUP during both resting and squeezing conditions. These findings suggest that tadalafil induce relaxation of urethral smooth muscle and the EUS (by means of the nitric oxide pathway). Furthermore, our data indicate that there is little or negligible PDE5 activity in the urethral vascular plexus, or that the net result of the simultaneous processes (in vascular smooth muscle and urethral smooth muscle and the EUS) is a mean decrease in urethral pressure, both during rest and voluntary contraction. In contrast, we found no statistically significant effect of tadalafil on voiding parameters. This might be explained by the fact that healthy women already have free flow. The observed period effect with increased flow rates at second visit is probably due to participants being more relaxed at the second uroflow measurement.
Concluding message
Compared to placebo, single-dose tadalafil decreased OUP during both resting and squeezing conditions in healthy women. No significant difference in voiding parameters were demonstrated. The observed fall in urethral pressure suggests a relaxing effect of PDE5 inhibition on EUS and urethral smooth muscle.
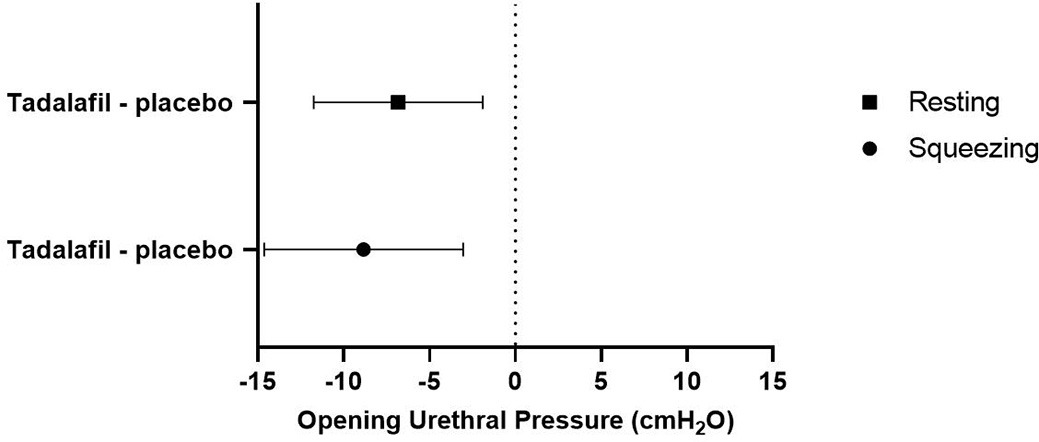
Figure 1. Mean opening urethral pressure at tmax corrected for the placebo value (mean OUP, cmH2O [95%CI]).
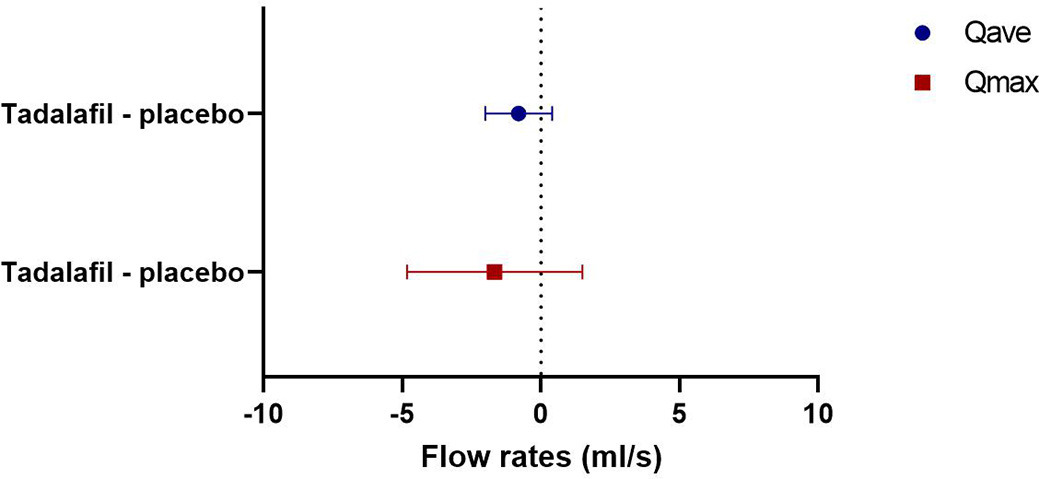
Figure 2. Mean flow rates (average flow rate [Qave] and maximum flow rate [Qmax]) corrected for the placebo value (ml/s [95%CI]).
References